The 9 Hallmarks of Ageing
Ageing is the progressive deterioration of an organism due to an accumulation of damage, resulting in structural and functional changes. This deterioration is the primary risk factor for several diseases including cancer, diabetes, cardiovascular disorders, and neurodegeneratve disease. To cure these diseases, we need to develop a better understanding of the ageing process and WHY it happens.
Scientists have identified nine hallmarks under 3 categories (primary, antagonistic, integrative) as common denominators of ageing in different organisms.
These hallmarks are genomic instability, telomere attrition, epigenetic alterations, deregulated nutrient sensing, mitochondrial dysfunction, cellular senescence, stem cell exhaustion, and altered intercellular communication.
The primary causes of ageing are triggers of age related decline with negative effects. Along with this, antagonistic hallmarks are beneficial effects of ageing just as long as they are not too abundant. Lastly, integrative hallmarks are a result of primary and antagonistic factors.

GENOMIC INSTABILITY (PRIMARY)
DNA in one of our cells gets damaged thousands of times a day. As we get older, this damage accumulates, and as a result, gene expression patterns are affected. DNA damage consists of alteration in the chemical or physical structure of DNA molecules. Damage can be a break in a DNA strand, a base missing from a strand, or anything that induces abnormalities in DNA molecules.
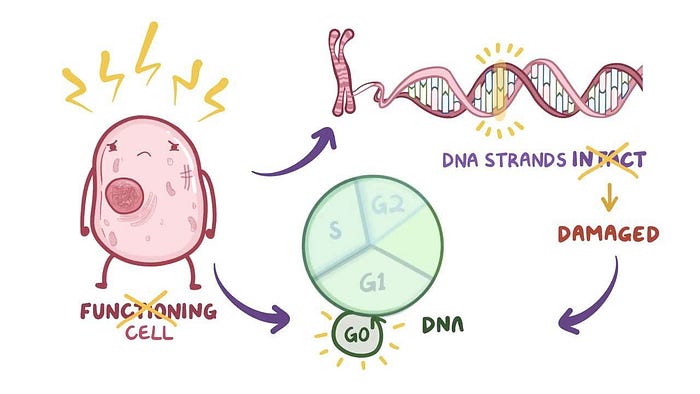
There are many consequences of a damaged genome, as it changes the way our cells work and causes genomic instability. The body’s failure to maintain homeostasis (remain stable regardless of the changes in internal and external cell conditions) is known to occur with ageing.
It’s important to note that DNA damage is different from DNA mutations. DNA damage is physical or chemical damage that occurs; whether something gets added or deleted in the genome, molecules become nonviable and are rendered useless within the cell. This is because replication, transcription, and DNA translation are halted, resulting in no flow of genetic information. DNA damage is followed by DDR (DNA damage response), which attempts to fix what has occurred due to any endogenous or exogenous cause. Our body’s DDR repairs most damages in DNA.
DNA mutations, on the other hand, occur when there is a physical or chemical change. Although they may differ from normal molecules or produce non-functional protein, the molecules still work, and there is no halt in processes. DNA repair is almost nonexistent when there are mutations, causing it to remain within the cell. Spontaneous mutations result from errors in natural biological processes, while induced mutations are due to agents in the environment that cause changes in DNA structure.
There are large-scale mutations where a whole chunk of the chromosome is lost, relocated, or rearranged. On the contrary, point mutations are a change in one base pair in a DNA molecule. A difference of one nucleotide can be enough to cause significant problems in the body. For example, sickle cell anemia occurs because of a difference in a single nucleotide in the DNA of a carrier compared to a non-carrier’s DNA. The difference in the gene that codes for a subunit of hemoglobin. Hemoglobin is a protein that carries oxygen through the bloodstream. Due to this point mutation, there is an “A” where there should be a “T” in the template strand of the gene which codes for “U” instead of “A” in the corresponding mRNA. This ultra codon will code for valine (an amino acid) instead of glutamic acid. In turn, this mutation causes hemoglobin to aggregate in low oxygen conditions, forming hemoglobin fibres. The fibres carry red blood cells that carry hemoglobin will be distorted into rigid, sticky sickle-shaped cells. These can get stuck in small blood vessels, which can slow or block blood flow and oxygen to parts of the body. Essentially, one point mutation can be disastrous.
DNA Damage and its causes occur by endogenous (internal) and exogenous (external) sources. These endogenous sources include ROS (reactive oxygen species), replication errors, inadequate repair systems, and incidental damage. Exogenous sources include UV Radiations, X-Rays, Gamma Rays, Hydrolysis, intercalating agents, and viruses.
ROS is an unstable molecule. It contains oxygen and easily reacts with other molecules in a cell. The build-up of ROS in cells may cause damage to DNA, RNA and proteins, also causing cell death. A theory of ageing results from oxidative damage by free radicals (ROS) on DNA, proteins, lipids leading to a loss of these molecules function. ROS have a role to play in many human diseases such as Parkinson’s disease, multiple sclerosis, muscular dystrophy, cataracts, Retinopathies, atherosclerosis, myocardial infarction, Ischemia and Reperfusion syndrome, pulmonary emphysema, hepatic cirrhosis, arthritis rheumatoid, and various types of cancer.
TELOMERE LENGTH (PRIMARY)
Our cells constantly undergo mitosis and meiosis to make copies of DNA packed into chromosomes. For every cell that undergoes division, a little bit of DNA is lost at the ends. This is why we have telomeres, repetitive nucleotide sequences that protect the ends of the chromosomes from degradation and fusion. As telomeres shorten, the integrity of the DNA is not maintained and can result in tumourigenesis. The division occurs until the telomeres are too short and can no longer divide, introducing cellular senescence.
Telomere length decreases with age, and as humans, the length of our telomeres seems to fall at a rate of 24.8–27.7 base pairs in a year. The length of one’s telomeres can be used as a predictor of biological age. Environmental and lifestyle factors such as smoking, obesity, unhealthy diet, lack of exercise, and harmful agents induce DNA damage, telomere shortening, increase cancer risk, and speed up the ageing process. Accelerated shortening of telomeres is seen to be associated with heart failure, diabetes, cancer risk, and osteoporosis.
EPIGENETIC ALTERATIONS (PRIMARY)
Epigenetics is the study of heritable phenotype changes that don’t involve alterations in the DNA sequence. The epigenome can be thought of as a layer on top of the traditional genetic genome sequences. Changes in the epigenome as you age alter gene expression and negatively affect the correct function of cells. These epigenetic changes are reversible; They don’t change your DNA sequence but change how your body reads a DNA sequence.
Lifestyle factors such as diet, obesity, physical activity, tobacco smoking, alcohol, psychological stress, and working night shifts have been identified to modify epigenetic patterns- weakening the immune system and contributing to the development of inflammation.
Environmental factors and pollutants can also cause metabolic errors leading to ageing, lowered adaptive capacity, and age-associated pathologies.
That being said, as genetically identical twins age, they can have significant differences in their genome methylation patterns, gene expression, and ultimately lifespan. Epigenetic changes account for 80% of the ageing process.
The causes of epigenetic changes can be attributed to DNA methylation, histone modification, and chromatin remodelling. These changes are responsible for human diseases such as Angelman’s syndrome, Prader-Willi Syndrome, Fragile X syndrome, and various cancers. An epigenetic clock is a better predictor of biological age than how old we are. It’s a biochemical test based on DNA methylation levels.
LOSS OF PROTEOSTASIS (PRIMARY)
Proteostasis is the regulation of a balanced, functional proteome (set of proteins expressed by a genome, cell, tissue, or organism at a particular time).
Loss of proteostasis is when the cells’ protein-building machinery fails, and there is an accumulation of misfolded proteins. These misfolded proteins are among the root causes of age-related diseases, the most significant one being Alzheimer’s disease. Functional proteins are correctly folded, but as you get older, the protein synthesis rate decreases and the protein degradation rate is reduced (the same amount of protein, it’s just been there for longer).
The longer it’s been, the more likely the protein is to aggregate. Aggregation is a crucial cause of ageing. Specifically, aggregation in amyloid-beta is the leading cause of Alzheimer’s disease.
Grifol’s completed a final phase clinical trial and managed to slow down Alzheimer’s disease by over 50%. They drew blood from the patient & cleaned it of waste, including Amyloid Beta, then injected it back into the brain.
Arethusa was another clinical trial being tested in animals and seems quite promising. They unclog a waste clearing passage and allows the older, diseased brain to maintain itself just like a young brain would.
TLDR: Alzheimer’s (a widespread age-related disease) happens due to an accumulation of 2 types of misfolded, damaged proteins.
- Amyloid beta (found in the spaces in between the cells)
- Tau protein (found in the cells)
The misfolded proteins can cause other proteins to transform into them on contact resulting in a catastrophic spread of damaged proteins. With Alzheimer’s disease, Amyloid-beta seems to help spread Tao.
Proteins are made of diff combinations of amino acids, following the exact blueprint given by the instructions of the DNA.
They do everything- maintain the structure of cells, regulate almost everything in the body, critical for function, regulation and structure of tissues and organs. The accumulation of damaged proteins due to glitches, misfolding, or sugar-binding to them turns them into harmful versions of themselves.
Loss of proteostasis occurs when damaged proteins build up and cause disease- whether it be Alzheimer’s, heart or another age-related disease.
CELLULAR SENESCENCE (ANTAGONISTIC)
The ageing of tissues is a result of the accumulation of senescent cells. When cells become senescent, they underproduce the protein that tells them when it’s time to die. To resist cell death, senescent cells activate pro-survival and inhibit pro-apoptotic pathways.
Senescent cells secrete pro-inflammatory cytokines. These cytokines activate nearby immune cells to fire away nasty chemicals, which damage more cells and cause inflammation, tissue dysfunction, and diabetic complication. The cytokines are produced from a phenotype called SASP as a pro-inflammatory response.
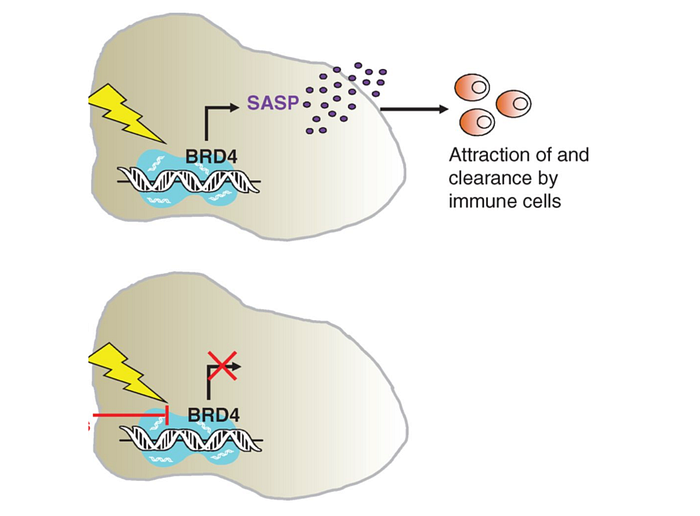
SASP disrupts normal tissue function in many ways:
- Accumulation of inflammatory immune cells.
- Remodelling of the extracellular matrix (which is the environment where cells are embedded)
- Induction of fibrosis.
- Decline of stem cell function
MITOCHONDRIAL DYSFUNCTION (ANTAGONISTIC)
Mitochondria is essential for respiration which enables energy production for the cell. Over time this process becomes less efficient.
Mitochondria produces ATP (adenosine triphosphate), used to store the energy required by the cell. When Mitochondria damage occurs, DNA can mutate, and proteins that are needed for mitochondria to function can be damaged by oxidation. Dysfunctional mitochondria produce less ATP, cutting down the energy supply of the cell. This gives off biochemical signals that induce cell death, and the wrong moment and leakiness can generate free radicals to damage a cell’s proteins and lipids. Mitochondria have an essential role to play in a cell and are a part of intracellular homeostasis, calcium balance, and metabolism processes. Also, Mitochondrial DNA mutates 10x faster than DNA in the nucleus and a lower capacity to repair DNA, playing a role in ageing and cancer. This powerhouse of the cell is recognized as one of the most critical factors of the ageing process, specifically in age-related neurodegenerative diseases.
DEREGULATED NUTRIENT-SENSING (ANTAGONISTIC)
This hallmark is concerns how our cells measure amount of nutrients available in the human body. Each cell can be thought of as a tiny factory full of molecular machines. For the factory to operate, it needs fuel and raw materials. We need to know how much fuel we have to know how hard the factory needs to work. Similarly, each cell need nutrients to operate, and the body needs to know how many nutrients we have in order to decide how hard the body is going to work. mTOR and IIS are pathways that detect high nutrient levels. AMPK and SIRT are molecular pathways used to detect low nutrient levels.
Let’s go deeper into mTOR- a molecular pathway that detects high nutrient levels and increases cellular metabolism in response. In a study it was shown that yeast, worms, and flies with mutated or deleted version of mTOR gene lived longer.
Scientists have found that they can also increase human lifespan by blocking mTOR activity with a drug called Rapamycin. When mice were fed Rapamycin along with their food, their life span was increased by up to 15% longer. This is projected to have a similar effect in humans. The study was done in older mice in age (equivalent to 60 year old person). Unfortunately, inhibiting mTOR can also have nasty side effects- so this is definitely not the holy grail solution.
The main idea is that cells monitor nutrient availability and regulate activity to balance growth and damage. The deregulation of this process plays a roll in the ageing process.
STEM CELLS EXHAUSTION (INTEGRATIVE)
Stem cells are cell blueprints sitting at various places in the body, copying themselves to produce new young cells. Stem cells are undifferentiated, which is why they’re used in regenerative medicine.

The problem is- our stem cell count declines as we age, leaving us less able to repair damage in the body.
They’ve done many great studies in mice concerning stem cells in longevity. As stem cells count decreased, the mice developed more diseases. Scientists took stem cells from baby mice’s brains and injected them directly into the brains of middle-aged mice. The new stem cells secreted RNA’s which regulated the mice’s metabolism and reinvigorated older brain cells.
After four months, the brain and muscle of these mice worked better, causing them to live 10% longer. In another study, stem cells from mice embryos were injected into the hearts of older mice. The result was improved heart function, ability to exercise 20% longer, and much faster regrowth of hair.
ALTERED INTERCELLULAR SIGNALLING (INTEGRATIVE)
To communicate with to each other, cells in your body use an intricate network of chemical signals. However, we don’t have a full understanding of this system yet. Altered intercellular signalling consists of gradual harmful changes to the chemical intercom cells use (which deteriorates as you get older).
The degradation of cell communication has a negative impact on how cells behave but also on their surrounding microenvironment, creating a negative spiral. This manifests in chronic inflammation. When the body undergoes infections and damaged cells, there is a positive inflammatory response. When the inflammation is chronic, it doesn’t solve problems- it makes things worse and can lead to obesity, atherosclerosis, and type 2 diabetes. Chronic inflammation also messes with your stem cell function, bone fragility, and muscle weaknesses.
Altered intercellular signalling is a common feature of ageing & product of hallmarks such as genomic instability (which leads to low efficiency of them) body’s repair system) and cellular senescence.
Primary hallmarks are all negative, consisting of DNA damage, mutations in mitochondrial DNA, telomere loss, epigenetic changes, and defective proteostasis. Antagonistic hallmarks on the other hand have beneficial effects (at a low intensity) but detrimental (at a high intensity). This is seen with cellular senescence which protects organisms from cancer but promotes ageing with high accumulation on senescent cells. Likewise, reactive oxygen species mediate cell signalling but at high chronic levels- can produce cellular damage. Lastly- integrative hallmarks are a combination of the other types of hallmarks, directly affecting tissue homeostasis and function.
